More on Water and Vapor Pressure
We continually deal with water in our business. Either it is a problem
that needs to be solved in the pump or a process involving water. It
many times revolves around the physical chemistry of water and its' various
phases. Gas, liquid, steam or ice. Water behaves like any other
liquid when we hit it with a vacuum the following explanations are meant to
impart some of this knowledge and how it might effect your own process or
problem. PV=nRT, Boyles Law, Charles Law with volume kept constant we can
simplify the equation often to P=T or pressure is inversely proportional to
temperature for most any liquids.
The boiling point is defined as the
temperature at which the
saturated vapor pressure of a liquid is
equal to the surrounding
atmospheric pressure. For water, the vapor
pressure reaches the standard sea level atmospheric pressure of 760 mmHg at
100 degrees C. Since the vapor pressure increases with temperature, it
follows that for pressure greater than 760 mm Hg (e.g., in a pressure
cooker), the boiling point is above 100C and for pressure less than 760 mmHg
(e.g., at
altitudes above sea level), the boiling
point will be lower than 100C. As long as a vessel of water is boiling at
760 mmHg, it will remain at 100C until the
phase change is complete. Rapidly boiling
water is not at a higher temperature than slowly boiling water. The
stability of the boiling point makes it a convenient calibration temperature
for
temperature scales.
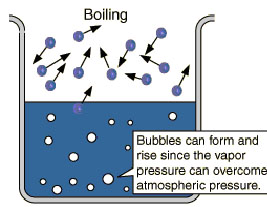
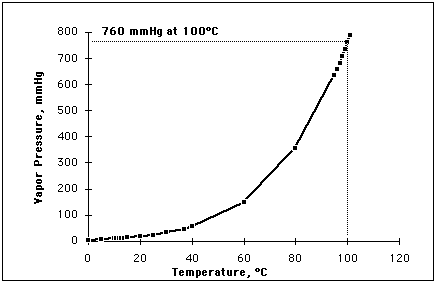
Remember 760 Torr or 760mm Hg if you will, is atmospheric pressure and
0 mm Hg is perfect vacuum. Therefore we can boil water at room
temperature
at roughly 5-10 Torr for talking sake. Same relationship, just
different numbers
for many other chemicals. You can imagine this might enable the user to
vary temperature and modify the presence or removal of water without using
necessarily higher temperatures which may degrade samples or effect yields. |
11/27/2005 ©Copyright HyVac Products, Inc. All Rights
Reserved
Toll Free 1-800-628-0850
|